Why This Matters
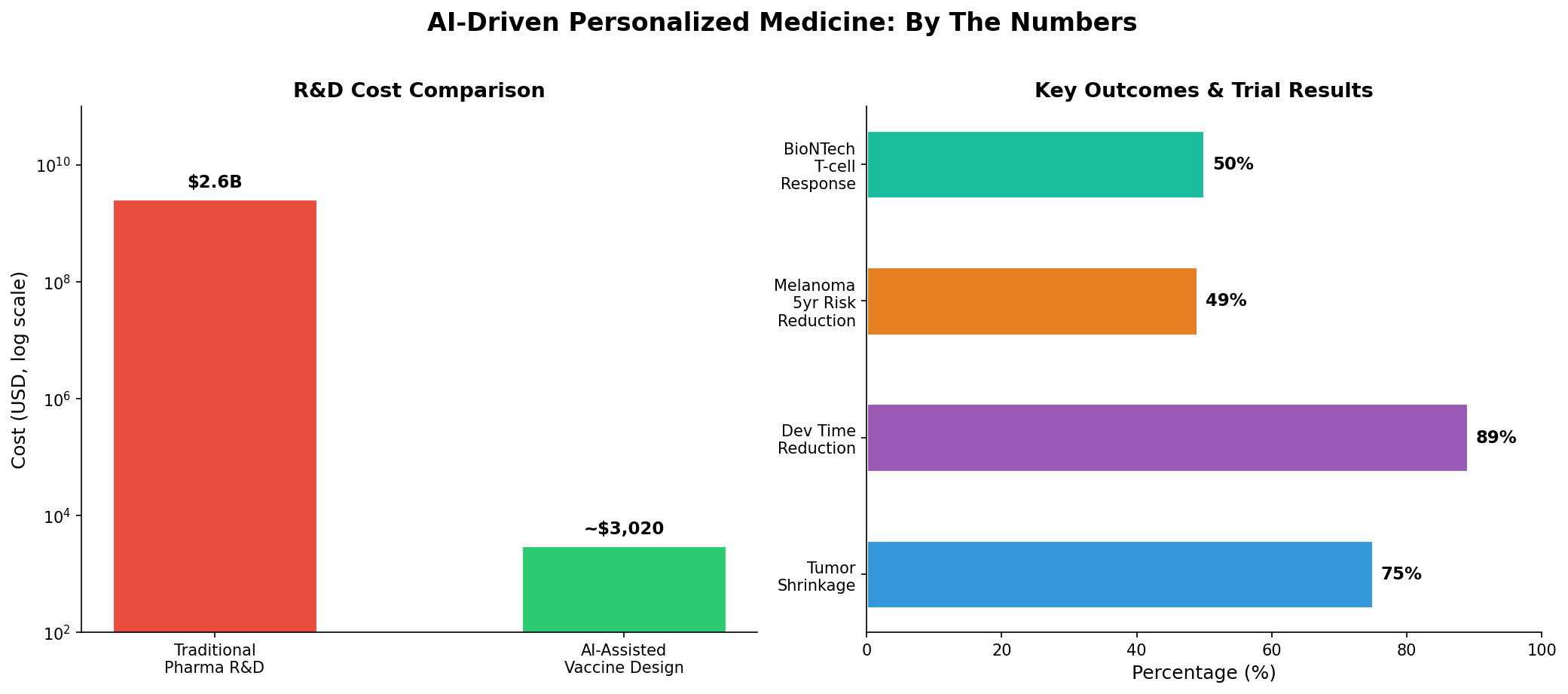
The story of Rosie the dog represents a potential inflection point in personalized medicine — not because a single animal was treated, but because of what the process reveals about the collapsing cost and complexity barriers in biomedical research. For decades, drug development has been a domain exclusively accessible to large pharmaceutical companies with billions in capital and years of runway. Paul Conyngham's AI-assisted approach, regardless of its clinical rigor, demonstrated that the core intellectual work of identifying neoantigens and designing vaccine candidates can now be performed by a technically literate individual using commercially available AI tools.
This matters because it previews a future where the rate-limiting step in personalized medicine shifts from discovery to manufacturing and regulation. The AI tools — ChatGPT for workflow guidance, AlphaFold for protein structure prediction, Grok for sequence design — are either free or available at consumer price points. The genomic sequencing cost $3,000, a fraction of what it would have cost even five years ago. The implications extend far beyond veterinary medicine: if the workflow proves replicable and safe, it could dramatically accelerate the development of personalized cancer vaccines for humans, particularly for rare cancers that lack commercial incentive for traditional pharma investment.